The Hidden Alchemy of Film / Plate Photography

At first glance, black-and-white photography feels simple. Light enters a camera, a plate or film is exposed, and in the darkroom an image slowly appears. But beneath that simplicity lies one of the most refined chemical systems ever developed, a process in which light quite literally rearranges atoms of metal.
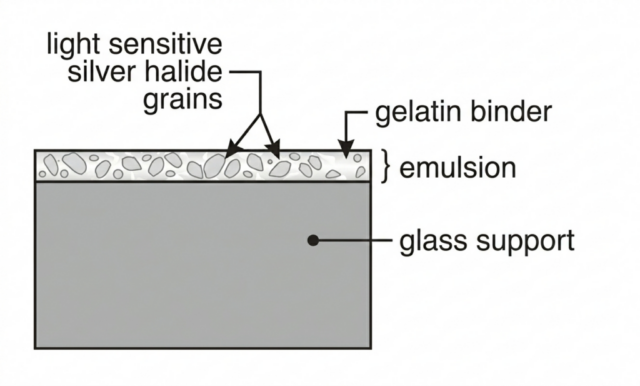
Every traditional black-and-white photographic plate, sheet film, roll film, and darkroom paper begins with the same essential structure: microscopic crystals of silver halide suspended in gelatin. These crystals are compounds formed between silver and halogen elements, most commonly bromine, sometimes chlorine or iodine. The result is silver bromide, silver chloride, or mixed halide crystals engineered for precise sensitivity and tonal control.
These crystals are astonishingly small. Many measure less than one micron in diameter. Hundreds could sit side by side across the width of a human hair. And yet, inside each of them lies the potential to record light with extraordinary precision.
The gelatin that holds them in suspension is not simply a binder. It is a carefully controlled environment. It allows chemicals to move during development, protects the crystals from physical damage, and provides long-term stability. The invention of the gelatin dry plate in the 1870s replaced earlier wet processes and transformed photography into a practical, portable medium.
Whether coated onto glass, flexible plastic, or baryta-coated paper, the chemistry remains fundamentally the same
Why Silver Halide Reacts to Light
The light sensitivity of silver halide is rooted in solid-state physics.
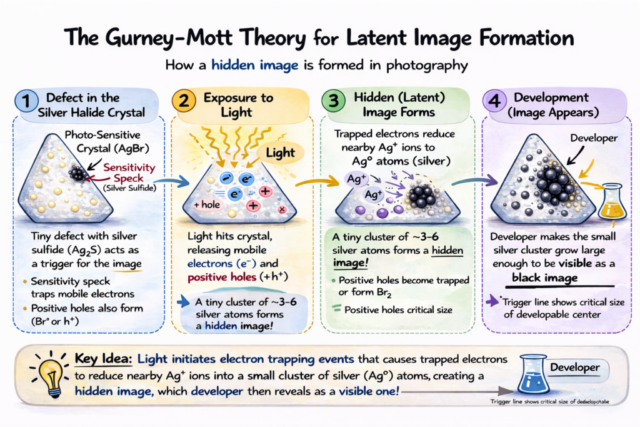
When a photon strikes a silver halide crystal, it transfers energy to an electron within the crystal lattice. That energized electron becomes mobile and migrates until it encounters a tiny imperfection known as a sensitivity speck. These specks are deliberately formed during emulsion ripening and are essential to photographic sensitivity.
At the speck, the freed electron reduces a silver ion to metallic silver.
One atom.
Then another.
Then another.
It takes only a small cluster, typically four or more silver atoms, to create what scientists call a stable latent image center. This cluster is far too small to be visible, but it marks the crystal as exposed.
Photography begins with changes occurring at the level of only a few atoms.
The plate still looks blank. But it has already been altered in a way that cannot be undone.
Exposure: Writing an Invisible Record
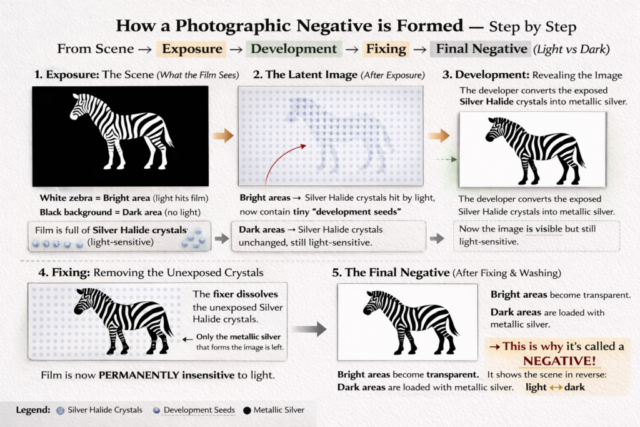
When a scene is exposed in the camera, bright areas send vast numbers of photons into the emulsion. Many crystals accumulate latent image centers. Dark areas send fewer photons and form fewer clusters.
At this stage, the material contains a precise atomic map of light intensity, but no visible image.
The latent image can remain stable for long periods under proper conditions. Historical accounts from polar expeditions in the late nineteenth century describe exposed plates that were developed years later and still produced images. The structure created by light can quietly endure until development reveals it.
The photograph exists before it is seen.
Development: Amplifying the Atomic Trace
The developer solution contains reducing agents that act preferentially on crystals containing latent image centers. The tiny cluster of silver atoms formed during exposure acts as a catalyst. The entire crystal is reduced to metallic silver.
This is chemical amplification on an extraordinary scale.
A cluster that may have begun with only a few atoms becomes a grain containing billions of silver atoms. Without this amplification effect, photography would require impossibly intense light. The darkroom transforms atomic traces into visible structure.
Where exposure was strong, many crystals convert to metallic silver. Where exposure was weak, few convert. Gradually, the image emerges in shades of gray and black.
What we are seeing is not tone in the abstract.
We are seeing density variations in metal.
The Negative: Reversal of Light
After development, the image appears reversed.
Bright areas of the original scene now contain dense accumulations of metallic silver and appear dark. Shadows remain relatively clear. This reversal is a physical consequence of silver blocking light. The more silver present, the less light passes through.
The negative is therefore a direct physical record of photon distribution translated into metallic density.
Fixing: Making the Image Permanent
Even after development, unexposed silver halide crystals remain embedded in the gelatin. These crystals are still light sensitive. If left in place, they would gradually darken and destroy the image.
Fixer removes them.
Sodium thiosulfate dissolves the remaining silver halide by forming soluble complexes, which are washed away. What remains is metallic silver, stable and no longer reactive to light.
Properly processed silver gelatine photographs have demonstrated remarkable longevity. Many prints from the nineteenth century remain intact today, their tonal scale preserved by the stability of metallic silver.
Grain: The Physical Structure of the Image
When photographers speak of grain, they are referring to the visible clumping of metallic silver particles formed during development.
Crystal size plays a critical role. Larger crystals generally provide higher sensitivity but produce more visible grain. Smaller crystals yield finer detail but require more exposure. Twentieth-century advances in crystal engineering, including tabular grain structures, improved efficiency while maintaining fine image quality.
Under magnification, a black-and-white photograph reveals itself as a landscape of metallic particles shaped by light.
From Negative to Positive
The negative is only an intermediate stage.
When light passes through a negative onto photographic paper, the tones reverse once more. Clear areas in the negative allow light to reach the paper, forming dark tones during development. Dense areas block light and produce lighter tones.
Once again, silver halide is reduced to metallic silver. Once again, fixer removes the unused salts.
The final print is composed of silver metal. Light has reorganized atoms twice, first to create the negative, then to create the print.
Why Silver?
Silver possesses a rare combination of properties. It forms light-sensitive halide compounds. These compounds undergo electron-based reactions when struck by photons. The reduced metallic silver is opaque and visually black in fine particles. Unexposed salts can be chemically removed without disturbing the metallic image. The final metallic structure is chemically stable under proper storage.
Few elements offer this combination of sensitivity, reversibility, and permanence. That is why silver has remained at the heart of photographic technology for nearly two centuries.
A Material That Records Light as Metal
When you hold a silver gelatine plate or negative up to the light, you are not looking at pigment or dye.
You are looking at billions of silver atoms rearranged by photons.
The highlights and shadows are variations in metallic density. The tones are differences in how much metal blocks or reflects light. The image is a physical structure built from matter transformed by energy.
Photography, in its classical form, is controlled metal formation triggered by light.
And every photograph is a small, permanent fragment of light turned into silver.